Efficiency is a measure of how much work or energy is conserved in a process. In many processes, work or energy is lost, for example as waste heat or vibration. The efficiency is the energy output, divided by the energy input, and expressed as a percentage. A perfect process would have an efficiency of 100%.

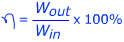
η = efficiency (Greek letter “eta”)
Wout = the work or energy produced by a process. Units are Joules (J).
Win = the work or energy put into a process. Units are Joules (J).
Efficiency Formula Questions:
1) A construction worker puts 20 J of energy into one strike of his hammer on the head of a nail. The energy transferred to driving the nail into the wood is 8.0 J. What is the efficiency of the construction worker’s hammering?
Answer: The efficiency can be found using the formula:
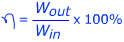
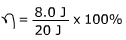
η = 0.40 x 100%
η = 40%
The efficiency of the hammer strike was 40%. Vibrations and heating of the nail are two possible reasons for the energy loss.
2) A particular chemical process has an energy efficiency of only 3.00%. To complete this large-scale chemical process, 140,000 J of energy is input. What is the energy output of this process?
Answer: The energy output can be found by rearranging the formula for efficiency:
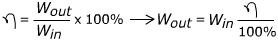
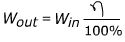
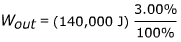
Wout = (140,000 J)(0.03)
Wout = 42,000 J
The energy output of this chemical process is 42,000 J.